Friday, September 01, 2006
Researchers Provide Evidence of How Proteins Fold
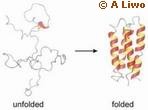 Experimental evidence provided by a Cornell researcher and colleagues at the Scripps Research Institute in La Jolla, California, support a long-held theory of how and where proteins fold to create their characteristic shapes and biological functions.
Experimental evidence provided by a Cornell researcher and colleagues at the Scripps Research Institute in La Jolla, California, support a long-held theory of how and where proteins fold to create their characteristic shapes and biological functions.
The theory proposes that proteins start to fold in specific places along an amino acid chain (called a polypeptide chain) that contains non-polar groups, or groups of molecules without a charge, and continue to fold by aggregation, i.e., as several individuals of these non-polar groupings combine. Using the same principle that separates oil and water, these molecules are hydrophobic - they avoid water and associate with each other.
--
Based on the Proceedings of the National Academy of Sciences (PNAS) paper "The role of hydrophobic interactions in initiation and propagation of protein folding" (Abstract).
technorati tags: cornell, scripps, research, institute, la+jolla, california, theory, proteins, fold, amino, acid, chain, polypeptide, non, polar, molecules, aggregation, hydrophobic, water, oil, proceedings, national, academy, sciences, pnas, protein, folding
Add to: CiteUlike | Connotea | Del.icio.us | Digg | Furl | Newsvine | Reddit | Yahoo